The Biologic Shift: Opening New Doors for Uncontrolled COPD Care

In the management of Chronic Obstructive Pulmonary Disease (COPD), the clinical community has long operated within a relatively defined framework of inhaled therapies. For many patients, the combination of bronchodilators and inhaled corticosteroids provides sufficient symptom control and a reduction in the frequency of exacerbations. However, there remains a significant cohort of individuals for whom standard therapy is not enough. These patients live in a state of "uncontrolled" disease, characterized by persistent symptoms and frequent, often debilitating, flare-ups that lead to emergency hospital admissions.
Recent developments in clinical guidance and the introduction of targeted biologic therapies mark a significant shift in this landscape. For the first time, the NHS is looking toward a precision medicine approach for COPD, specifically targeting patients with evidence of eosinophilic inflammation. This shift is not just about a new treatment option; it is about the evolution of the respiratory pathway and the potential to fundamentally change the way we manage high-risk patients across the system.
Understanding the Eosinophilic Phenotype
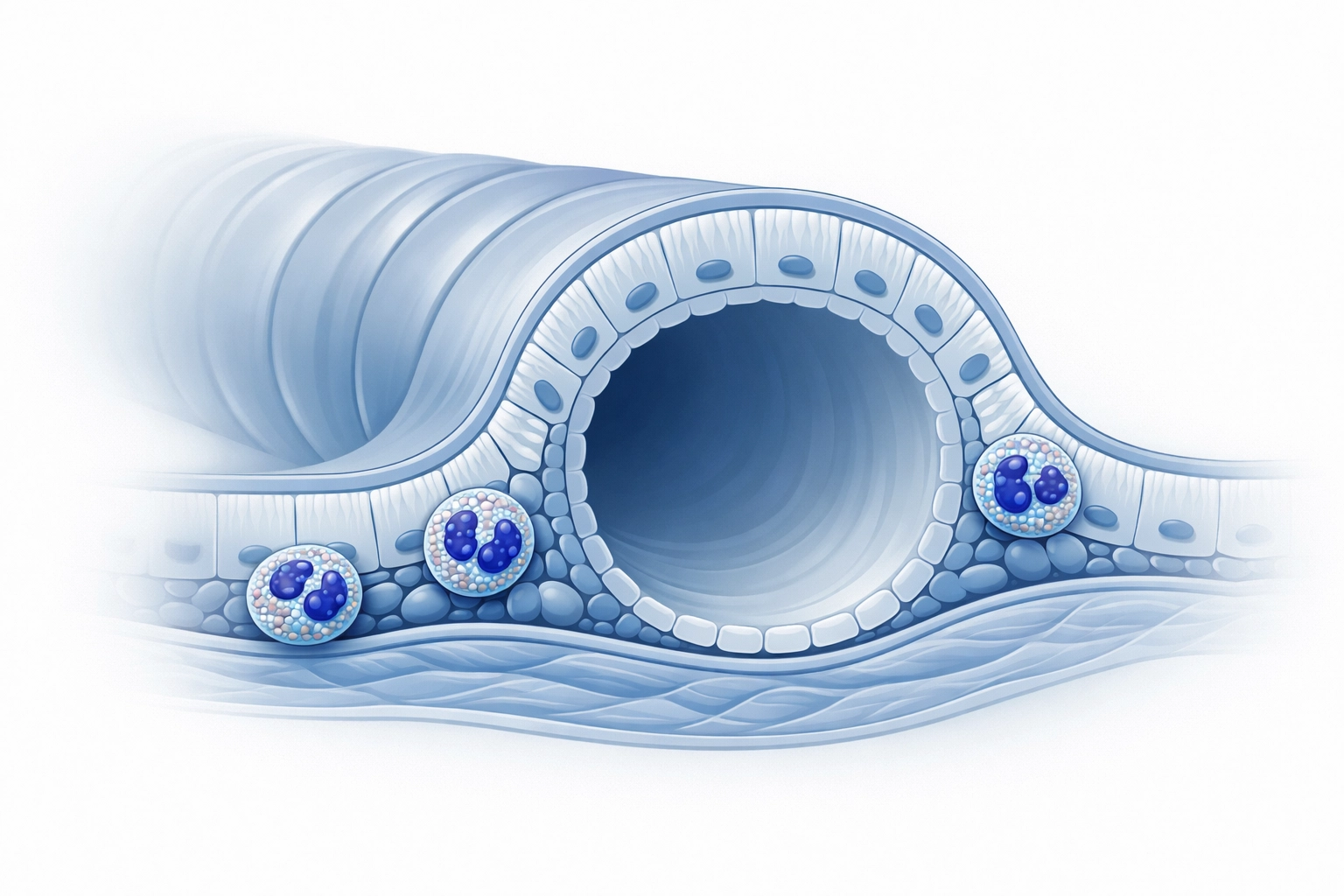
For years, COPD was often viewed as a singular, progressive condition. However, our understanding of the underlying biology has become increasingly sophisticated. We now recognize that COPD is heterogeneous, with different biological drivers: or endotypes: contributing to the clinical presentation.
One of the most significant insights in recent years has been the role of Type 2 inflammation, often identified by elevated blood eosinophil counts. In a subset of COPD patients, this specific inflammatory pathway remains active despite the use of maximal inhaled therapy. For these individuals, the "one-size-fits-all" approach to treatment frequently fails. They remain at high risk of exacerbations, which not only diminish their quality of life but also cause cumulative lung damage and place a heavy burden on NHS urgent care services.
The introduction of biologic therapy specifically designed to modulate these inflammatory pathways offers a new level of clinical precision. By addressing the root cause of the inflammation rather than just treating the symptoms, these therapies provide an alternative for those who have previously had very few options left on the table.
The System Impact: Reducing the Burden of Flare-Ups
From a system leadership perspective, the primary driver for integrating biologics into the COPD pathway is the reduction of exacerbations. In practice, a "flare-up" is rarely just a temporary worsening of symptoms. For the patient, it represents a period of acute distress and a potential loss of independence. For the NHS, it represents a predictable yet often preventable pressure on hospital beds and primary care appointments.
Recent NICE guidance has recognized the value of targeted intervention for those with uncontrolled COPD and high eosinophil counts. By identifying these patients early and moving them onto a biologic pathway, the goal is to stabilize their condition before they reach the point of crisis. The data suggests that reducing moderate to severe exacerbations can have a profound impact on hospital occupancy rates and the overall cost of care within an Integrated Care System (ICS).
What this looks like on the ground is a move toward more proactive identification. Clinicians are now looking more closely at blood eosinophil levels as a key biomarker to guide treatment escalation. This shift requires a high degree of coordination between primary care, where many of these patients are first managed, and secondary care respiratory specialists who oversee the administration of these advanced therapies.
Pathway Innovation and the Role of the ICS
The implementation of new clinical guidance is rarely a straightforward task. It requires a rethink of the existing respiratory pathway to ensure that the right patients are identified and referred at the right time. In the context of the NHS Long Term Plan and the development of Integrated Care Systems, this is an opportunity for genuine pathway innovation.
An effective biologic pathway for COPD involves several key stages:
- Identification: Using existing primary care data to identify patients who are frequently exacerbating despite triple therapy.
- Diagnostics: Ensuring that blood eosinophil counts are recorded and reviewed as part of the annual respiratory check or post-exacerbation follow-up.
- Specialist Assessment: Creating clear referral routes to multidisciplinary teams (MDTs) who can assess eligibility for biologic therapy.
- Ongoing Monitoring: Establishing protocols for long-term review to ensure the treatment is delivering the expected benefits in lung function and quality of life.
This collaborative approach ensures that innovation doesn't happen in a vacuum. It requires Life Sciences partners to engage deeply with NHS leaders to understand the local pressures and workforce constraints that might impact the rollout of these new treatments.
Challenges in Clinical Practice
While the clinical evidence for biologic therapy in COPD is compelling, its introduction brings practical challenges that the respiratory community must address. One of the most common concerns described by clinicians is the "diagnosis backlog." The ability to accurately phenotype a patient relies on consistent diagnostic standards, including high-quality spirometry and timely blood tests.
Furthermore, there is the question of workforce capacity. Managing a biologic pathway requires specialist nursing and pharmacy input to coordinate the delivery of treatment and patient education. In an overstretched system, finding the "room" for these new processes is a frequent point of discussion at our regional round tables.
However, the consensus remains that the long-term benefits of keeping patients out of the hospital far outweigh the initial hurdles of implementation. By reducing the revolving door of emergency admissions, the system can eventually free up capacity for more proactive, preventative care.
A New Chapter for Life Sciences Engagement
The shift toward biologics in COPD also changes the nature of engagement between the NHS and the Life Sciences sector. It is no longer about simply providing a product; it is about partnering on the pathway. Industry experts are increasingly involved in supporting the NHS with data insights, helping to map patient populations and identifying where the greatest unmet need lies.
This collaborative professional setting is essential for the successful adoption of innovation. When Life Sciences, NHS leadership, and clinical experts work together, the focus remains on the system's goals: improving patient outcomes and ensuring sustainability in care.

The Patient Perspective: Lived Experience and Targeted Care
For the person living with COPD, the transition to a biologic therapy can represent a major turning point. Many patients who fall into the "uncontrolled" category have spent years feeling that their condition is inevitably declining. They often describe a sense of fear regarding the next exacerbation and the impact it will have on their ability to perform daily tasks.
Moving toward a targeted treatment plan provides more than just clinical improvement; it provides a sense of agency. When a patient understands that their treatment is being tailored to their specific biological profile, it often leads to better engagement with their overall care plan, including pulmonary rehabilitation and smoking cessation.
Conclusion: Looking Ahead
The biologic shift in COPD is a testament to the progress being made in respiratory medicine. It marks the transition from broad-spectrum management to a more nuanced, individualized approach. As NICE guidance continues to evolve and more data becomes available, the integration of these therapies will likely become a standard part of the respiratory pathway for the most vulnerable patients.
Success in this area will depend on our ability to communicate across the system. We must share insights on what works, address the barriers to implementation, and remain focused on the common goal of reducing the burden of respiratory disease in the UK.
Medical Disclaimer: The information provided in this blog is for informational purposes only and does not constitute medical advice. Always seek professional clinical guidance for medical conditions.
Join the Conversation
How is your local ICS approaching the implementation of these new pathways? Are you seeing a change in how patients with eosinophilic inflammation are identified in your practice?
We invite you to join our Members' Community to discuss these developments with peers across the NHS and Life Sciences. Together, we can shape the future of breath.

Category: Life Sciences & Innovation
Tags: COPD Care UK, NICE Guidelines, Pathway Innovation, NHS Strategy, Life Sciences Engagement UK







Responses